22 Mar Łukasiewicz-IMN is part of the TETALEAD project increasing raw material efficiency in zinc production
PROMETIA member Łukasiewicz-Institute of Non-Ferrous Metals (IMN) participates in the TETALEAD project which is developing a new hydrometallurgical concept with a low carbon footprint to increase raw material efficiency in zinc production. For this purpose, significant amounts of lead, silver and tin contained in the currently unrecovered leach residues are recycled.
The aim of the recent research project was to recover lead from zinc industry waste using an aqueous solution of an aliphatic polyamine (triethylenetetramine, TETA). It was assumed that the lead would be recovered with maximum yield and then lead carbonate, the main product, would be produced. Finally, the leach solution was regenerated to meet the criteria of a low-emission and waste-free process. The lead carbonate concentrate produced can be smelted into lead metal, which has the advantage over conventional lead sulphate smelting that the smelting temperature is lower, energy consumption is lower and less CO2, NOX and SO2 are emitted. The technology developed, using triethylene tetramine solution as the extracting agent, allows 85-90% lead to be recovered within 1-1.5 hours at ambient temperature.
A pilot installation was prepared at the Center of Hydroelectrometallurgy, at ŁUKASIEWICZ–Institute of Non-Ferrous Metals in Gliwice, Poland. More details about the setup can be seen and read below:
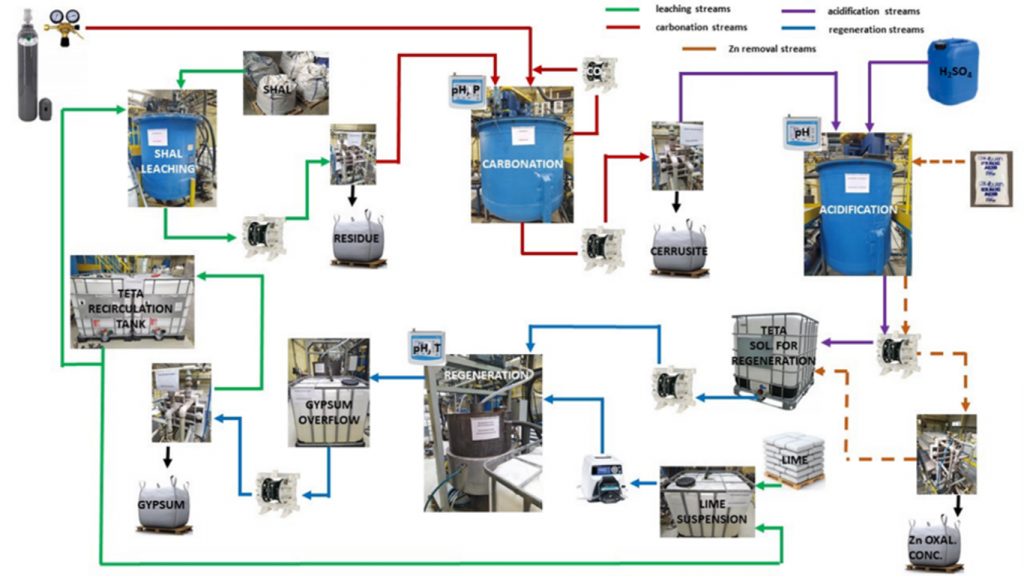
Schematic diagram of the pilot installation


Images of the pilot installation (hydro and pyro) 
THE SETUP
The technological line for the pilot plant consisted of a series of reactors for leaching, carbonation, acidification, Zn removal and regeneration with volumes of 1 m3, 2.8 m3, 1.7 m3 and 0.4 m3, respectively. The transport and pumping of all liquid streams was done in a closed loop, which is consistent with the low-emission and waste-free nature of the proposed technology. One pilot test used 4.4 tonnes of material and produced 730 kg of lead carbonate with an average moisture content of 10.8%, consuming 470 kg of CO2. The lead carbonate produced was then used in a pilot smelter to produce pure lead metal.
The pure lead carbonate was obtained, with a main contaminations being: Zn (<0.2%), Mg (<0.005%), Mn (<0.005%), and Ca (<0.3%). This product was a very good raw material for the production of crude lead with a good purity while generating a small amount of slag and low emissions of pollutants (SO2, NOx, CO) into the atmosphere. Membrane technique was proposed to recover amine from the circulating solution.




